5 Red Flags When Choosing a Research Peptide Supplier
The research peptide market has grown significantly in recent years. With that growth has come an influx of new suppliers, some operating to high standards, others with more variable standards. For researchers who depend on compound integrity for meaningful results, choosing the wrong supplier isn’t just a financial mistake. It can invalidate months of work.
Here are five red flags that should give any serious researcher pause before placing an order.
1. Pricing That Defies the Economics of Quality
High-purity peptides are expensive to produce correctly. When you see a supplier offering research-grade compounds at prices significantly below typical market ranges, it is worth asking what has been adjusted to make that price point possible.
Consider what genuine quality costs: manufacture under verifiable quality systems, independent third-party testing by an accredited laboratory, proper lyophilisation, controlled storage, compliant packaging, and documented chain of custody. None of this is inexpensive.
In many cases, suppliers offering unusually low prices while claiming high purity and third-party verification may indicate one or more of the following: testing that is less rigorous than represented, sourcing from unvetted manufacturers, underdosing of vials, or an unsustainable cost structure.
Researchers should ask: if the margins do not credibly support legitimate quality control, where are the standards most likely being compromised?
Pricing in the research peptide space should reflect the genuine cost of verified quality. When it does not, it is worth investigating what that gap represents.
2. Selling GLP-1 Receptor Agonists Alongside Research Peptides
GLP-1 receptor agonists, including compounds such as semaglutide and tirzepatide, have been approved as pharmaceutical drugs by regulators including the FDA and equivalent international bodies. Their status as approved pharmaceuticals creates a complex regulatory environment that differs significantly from typical research use only (RUO) peptides.
While GLP-1 compounds can exist in legitimate research contexts such as, preclinical studies or analytical reference standards, their sale through RUO channels is an area of active regulatory scrutiny and legal complexity that goes well beyond the framework applicable to most research peptides.
Suppliers who stock GLP-1s alongside conventional research peptides may reflect differing interpretations of regulatory frameworks or a higher tolerance for regulatory risk than many researchers would find acceptable. The RUO framework exists to support legitimate scientific inquiry. Suppliers who operate at its outer boundaries or beyond them can create risks that extend to their customers.
As a researcher, your supplier’s regulatory positioning is relevant to your own.
3. Permanent “Sale” Pricing and Constant Discounts
A supplier running a perpetual sitewide discount — 20%, 30%, or more, always active — is typically employing a pricing tactic rather than offering genuine value.
This matters for two reasons.
First, it suggests the “original” price is artificial. If a product is always discounted, the promotional price is effectively the real price. The inflated figure exists primarily to create the impression of a deal.
Second, and more relevant to researchers: in many cases, suppliers who compete primarily on price signal where their investment priorities lie. Rigorous procurement, accredited third-party testing, and documented quality control carry real costs. Where those costs are not reflected in pricing, it is reasonable to ask whether they are being absorbed or avoided.
Responsible research suppliers tend to price their products to reflect genuine cost of quality, and hold that pricing consistently. Stability in pricing can be a signal of stability in standards.
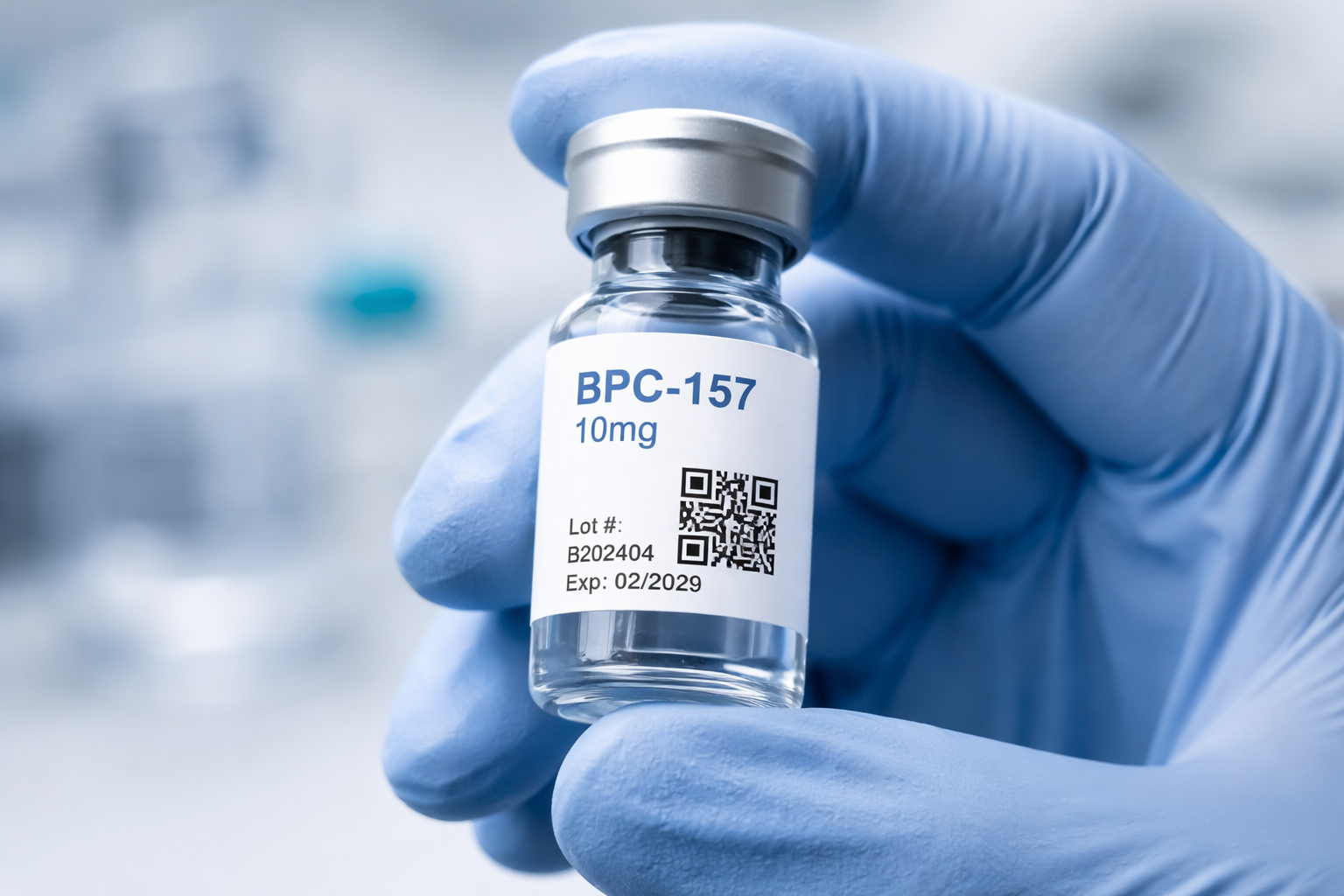
4. No Lot-Specific Certificate of Analysis Access
A Certificate of Analysis (COA) is the foundational document of peptide quality assurance. It should include compound identity, purity percentage, testing methodology, lot number, and the accreditation details of the testing laboratory.
But where and how a supplier provides COA access matters as much as whether they provide it at all.
A COA linked generically from a website without lot-specific traceability provides limited assurance. What researchers need is the ability to verify that the specific vial in hand corresponds to a specific tested batch with documented results.
A strong verification approach includes on-vial identification: a QR code or unique identifier printed directly on every vial that links to that batch’s COA. This creates a traceable chain from manufacture to the laboratory, and makes document misrepresentation significantly more difficult, a relevant consideration in a space where quality fraud does occur.
When evaluating a supplier, ask: can I verify the specific vial I am holding against documented test results, or am I relying on a document that could apply to any batch?
5. Reconstitution Supplies Sold Alongside Peptides
Bacteriostatic water, syringes, and reconstitution kits sold by a research peptide supplier, particularly when presented alongside peptides in a way that suggests combined use, can be interpreted as facilitating a specific end use that sits outside the intended scope of in vitro laboratory research.
The RUO classification is built on the premise that compounds are purchased for legitimate scientific research purposes. Suppliers who also provide the means of injection, in a context that implies that use, may find themselves — and their customers — in a more complex regulatory position than a straightforward RUO transaction would suggest.
Responsible RUO suppliers understand this distinction. Their products, documentation, and communications reflect the genuine boundaries of the RUO framework rather than testing its edges.
What to Look For Instead
The research peptide space does include suppliers who operate to high standards. They tend to share common characteristics:
Transparent, stable pricing that credibly reflects the cost of quality
- Lot-specific COA access tied to individual vials or documented batch numbers
- Accredited third-party testing – recognised analytical methods such as HPLC and mass spectrometry
- Clear RUO scope – no products or ancillary supplies that imply or facilitate injection use
– Documented and traceable manufacturing standards with verifiable supply chain
– Consistent standards not dependent on promotional pricing to attract buyers
Choosing a research peptide supplier is ultimately a quality control decision. The compounds you use shape the reliability of your results. Suppliers who understand that invest accordingly in verification, documentation, and compliance.
—
Varalion supplies research peptides independently verified by HPLC and Mass Spectrometry. Every vial carries a QR-linked Certificate of Analysis for lot-specific batch verification. For research use only. Must be 21 years of age or older to purchase.